Français / Español / 简体中文 / русский / العربية / 日本語
© 2026 World Federation of Hemophilia
For many years, people believed that only men could have symptoms of hemophilia and that women who “carry” the hemophilia gene do not experience symptoms themselves.
We now know that many carriers do experience symptoms of hemophilia. As our knowledge about the disorder has increased, so has our understanding of why and how women can be affected. Some women live with their symptoms for years without being diagnosed or even suspecting they have a bleeding disorder. Through education and awareness-raising, the World Federation of Hemophilia is working to close this gap in care.
Hemophilia is usually inherited, meaning that it is passed from parent to child through the parent’s genes. Genes are packaged within the body’s cells on structures called chromosomes. The genes involved in hemophilia are located on the “X” chromosome.
The X chromosome is also called the “sex chromosome” because it plays a role in determining whether a person is male or female. Men have one X chromosome, which they inherit from their mother, and one Y chromosome, which they inherit from their father. Women have two X chromosomes: one from each parent.
If the X chromosome a man inherits from his mother has the altered or changed gene, he will have hemophilia. If a woman inherits a copy of the altered gene from either of her parents, she is said to “carry” the hemophilia gene and is therefore called a “carrier”. In other words, she has one normal and one altered copy of the gene.
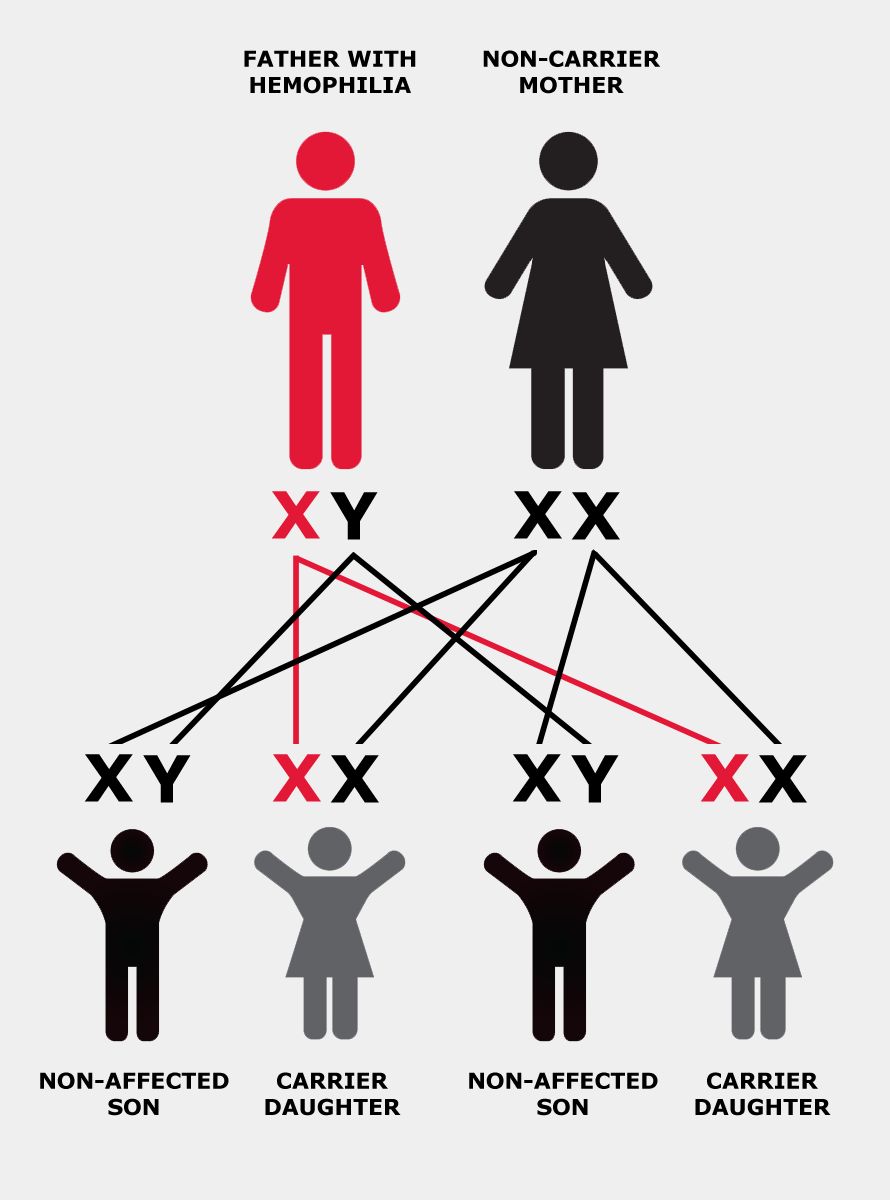
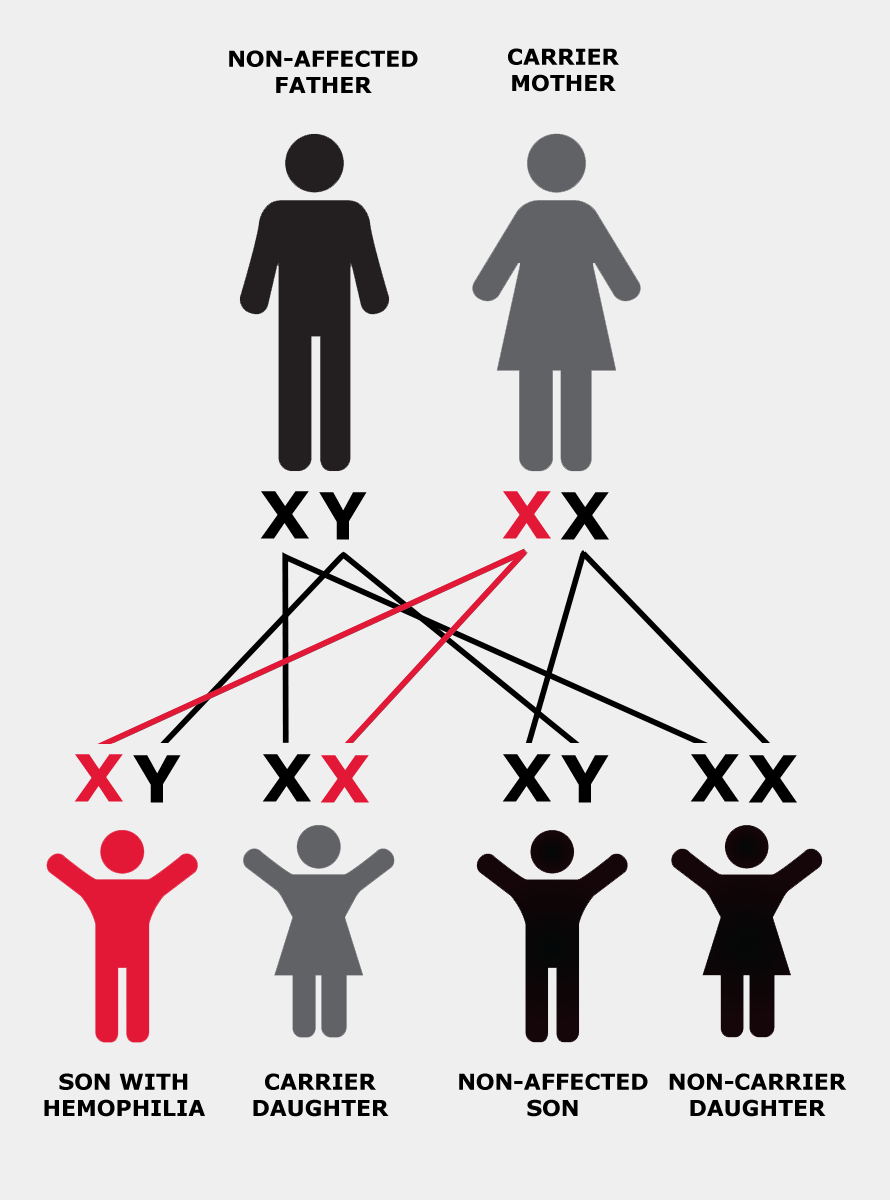
In each cell in a woman’s body, one of the two X chromosomes is turned off, or “suppressed”. This process is called “lyonization”, after Mary Lyon, who first described it. Lyonization is a random process that is not fully understood.
If the chromosome that’s turned off has the altered gene, that cell will produce clotting factor. If the chromosome with the normal gene is turned off, the cell will not produce clotting factor, or the clotting factor it makes won’t work properly.
On average, carriers of hemophilia will have about 50% of the normal amount of clotting factor, because about half of their cells will have the “good” gene turned off. Some carriers have far lower levels of clotting factor because more of the X chromosomes with the normal gene are switched off.
There are three levels of severity of hemophilia: mild, moderate, and severe. The severity of hemophilia depends on the amount of clotting factor in the person’s blood.
Any person (male or female) with 5%–40% of the normal amount of clotting factor has mild hemophilia.
A person with between 1%–5% of the normal amount of clotting factor has moderate hemophilia.
A person with less than 1% of the normal amount of clotting factor has severe hemophilia.
A woman who has less than 40% of the normal level of clotting factor is no different from a man with the same factor levels—she has hemophilia.
Some carriers have symptoms of hemophilia even though their clotting factor levels are above 40%. A woman with levels of 40%–60% of the normal amount of clotting factor who experiences abnormal bleeding is called a symptomatic carrier.
Approximately one third of carriers have clotting factor levels of less than 60% of normal and may experience abnormal bleeding. In most cases, carriers experience symptoms similar to those seen in men with mild hemophilia, as well as some that are specific to women, such as prolonged or heavy menstrual bleeding.
Symptomatic carriers and women with hemophilia:
Studies have shown that the most common symptom women experience is prolonged bleeding after surgery such as a tooth extraction or tonsillectomy. They are also at risk of serious bleeding after an accident or injury.
Carriers with low factor levels have a greater risk of heavy or prolonged menstrual bleeding (menorrhagia). Girls may have especially heavy bleeding when they begin to menstruate. Excessive blood loss can lead to anemia due to low levels of iron in the blood, resulting in weakness and fatigue.
Women with bleeding disorders are more likely to experience pain during their menstrual bleeding (dysmenorrhea). They may also experience a small amount of internal bleeding during ovulation, which can cause abdominal and pelvic pain (known as Mittelschmerz, a German word meaning “middle pain”). This bleeding can be severe or even life-threatening, especially in carriers with very low clotting factor levels, and may require urgent medical attention.
Menopause is the time in a woman’s life when menstrual periods permanently stop. Perimenopause is the 3- to 10-year period before menopause when hormones are “in transition.” Heavy and irregular menstrual bleeding occurs more commonly in all women as they approach menopause. Gynecological conditions (such as fibroids, polyps, etc.) are also more common at this stage of life. Carriers of hemophilia are at risk of more severe bleeding symptoms and may require treatment.
During the process of ovulation, women can develop simple (also called functional) ovarian cysts. These cysts are usually small, do not cause any problems, and go away on their own. Carriers of hemophilia seem to have a greater risk of bleeding into these simple cysts, which then become “hemorrhagic” ovarian cysts. Hemorrhagic ovarian cysts can cause considerable pain and may require urgent medical intervention.
Some carriers also suffer from endometriosis, a painful condition in which endometrial tissue, the tissue that lines the uterus, forms in the abdomen or other parts of the body. Although we do not yet understand the cause of endometriosis, women who experience heavy menstrual bleeding are more at risk of developing the condition.
There are two types of carriers: obligate carriers and possible carriers. Obligate carriers necessarily have the hemophilia gene, which they inherit from their father. Obligate carriers can be identified by getting a detailed family history (known as a pedigree).
| Obligate carriers are: | Possible carriers are: |
|---|---|
|
|
Two kinds of laboratory tests can be performed for carriers of hemophilia:
Diagnostic testing for carriers is a complex issue. While it is important for safety reasons to know a suspected carrier’s factor levels, genetic testing raises a number of ethical and cultural concerns.
Since carriers can be at risk of bleeding following trauma, tooth extractions, or other surgeries, it is a good idea to have factor levels measured in all suspected or known carriers so that extra precautions can be taken if factor levels are low. However, factor levels alone cannot confirm a woman’s carrier status.
The decision to undergo genetic testing is shaped by the family’s perceptions and cultural concerns, but also whether testing is accessible and/or permissible by regulatory bodies (i.e. government, insurance providers). In some countries, only the woman herself can consent to genetic tests; it is not a decision her parents can make for her.
Where genetic testing is possible before the child reaches the age of consent, families often struggle to determine when to test for carrier status. Many wonder whether they should have their daughters tested during childhood, specifically before they begin menstruating, or wait until they are adults and can make the decision themselves. Where possible, testing should be performed before a suspected carrier becomes pregnant.
Some families delay testing as a form of denial, or to protect the child and themselves from what they perceive as bad news. Cultural issues, such as arranged marriages or the possibility of the daughter having an affected child of her own, may discourage some families from having a daughter tested. Others test routinely as a matter of course, letting the child grow up with the knowledge of their carrier status. Knowing their status early can also help girls come to terms gradually with the complex reality of being a carrier.
In all cases, families should consult with the specialists at a hemophilia treatment centre or a genetic counsellor who can help them through the decision process and offer follow-up counselling, if necessary.
Some people simply accept the possibility of having a child with hemophilia. In countries where quality care with safe clotting factor concentrates is available, hemophilia is often seen as a manageable disease. Where adequate care is not available, this is a more difficult decision. Some families choose to adopt or foster a child, or to use other conception options to eliminate the risk of having a child with hemophilia. However, these options are not always available or can be unacceptable for religious, ethical, financial, or cultural reasons.
| Procedure | How it’s done | Things to consider |
|---|---|---|
|
In-vitro fertilization (IVF) with pre-implantation diagnosis (PGD) |
The woman’s eggs are retrieved and fertilized in the laboratory with the sperm from the woman’s partner. This is called IVF. When the embryos are at a very early stage of development, a test is done to determine whether they carry the altered hemophilia gene. Only those that do not contain the altered gene are implanted into the mother’s womb. |
This procedure is expensive and not available in many parts of the world. The success rate for a pregnancy with IVF is approximately 30% per cycle. CVS or amniocentesis is still recommended to confirm that the fetus does not carry the altered gene. |
| IVF with egg donation | Using donor eggs from a fertile woman who is not a carrier of hemophilia ensures that the child would not be at risk of inheriting the hemophilia gene from the mother. | Again, IVF is expensive, with a success rate for pregnancy of approximately 30% per cycle. The success rate is best when the donor is young. |
| Sperm sorting |
Only sperm carrying an X chromosome is used. This ensures the birth of a female child. |
The female child may still inherit the altered gene and have hemophilia. She could experience bleeding symptoms and may pass the altered gene on to her children. This method is only available in specialized centres as a research tool, and it is still under evaluation. |
Couples who have conceived naturally may wish to know whether their child is affected by hemophilia before he or she is born.
A definitive prenatal diagnosis can only be offered with invasive procedures such as amniocentesis or chorionic villus sampling. Some centres only offer these procedures if the couple plans to terminate the pregnancy if the fetus is found to have hemophilia. The decision to terminate a pregnancy is an extremely difficult one, which may not be acceptable for personal, religious, ethical, or cultural reasons.
Amniocentesis: A small amount of amniotic fluid is removed, using a fine needle inserted into the uterus through the abdomen. Amniocentesis is carried out under ultrasound guidance, between the 15th and 20th week of pregnancy. The amniotic fluid contains cells from the fetus that can be analyzed to detect hemophilia.
Chorionic villus sampling (CVS): Under local anesthesia and ultrasound guidance, a fine needle is inserted through the abdomen or a thin catheter is inserted through the mother’s vagina to take a sample of chorionic villi cells from the placenta. These cells contain the same genetic information as the fetus itself and can be used to determine whether the fetus is affected by hemophilia. This procedure is performed early—between the 11th and 14th weeks of pregnancy. CVS is the most widely used method for the prenatal diagnosis of hemophilia and other inherited bleeding disorders.
The risk of miscarriage associated with CVS or amniocentesis is up to 1%.
Pregnant women may also be offered non-invasive testing to determine the sex of the fetus they are carrying. This is done through testing the mother’s plasma for fetal cells that contain fetal DNA.
Fetal sex determination, which is finding out whether the baby is a boy or girl, is a relatively simple procedure. Knowing the sex of the fetus does not tell you if it is affected by hemophilia, but it does provide useful information.
If the fetus is male, and therefore has a greater likelihood of having hemophilia, CVS or amniocentesis can be offered to parents who wish to know whether he is affected with hemophilia. If the parents choose not to have CVS or amniocentesis, or if these tests are not available, doctors should plan labour and delivery to minimize the chance of bleeding in a male fetus (see “Labour and delivery: considerations for mother and child”).
If the fetus is female, prenatal diagnosis is not necessary because, even if the female child has hemophilia, there is very little risk of bleeding for the baby during labour and delivery.
The sex of the fetus can be determined in two ways:
Most women with hemophilia or hemophilia carriers have normal pregnancies without any bleeding complications. Levels of factor VIII increase significantly in pregnancy, which reduces the risk of bleeding for hemophilia A. Levels of factor IX, however, do not usually change significantly.
Factor levels should be tested in the third trimester of pregnancy, when they are at their highest. If levels are low, precautions should be taken during labour to reduce the risk of excessive bleeding. Women with hemophilia are at risk for postpartum hemorrhage, not only those with low levels but also those with normal levels of FVIII/IX. Postpartum hemorrhage occurs in approximately 20% to 50% of deliveries.
The obstetrician should work closely with the members of the hemophilia treatment centre to make sure the pregnancy is properly managed.
Planning for childbirth depends on the needs of the mother and her potentially affected child.
It is difficult to measure clotting factor levels during labour, so this should be done in the last trimester of pregnancy. If factor levels are low, treatment may be given during labour to reduce the risk of excessive bleeding during and after childbirth. Clotting factor levels may also determine whether a woman can receive local anesthesia (an epidural).
There is an increased risk of head bleeding in affected male babies, especially if the labour and delivery have been prolonged or complicated. Carriers may give birth vaginally, but prolonged labour should be avoided and delivery should occur in the least traumatic way possible. Invasive monitoring techniques such as fetal scalp electrodes and fetal blood sampling should be avoided whenever possible. Delivery by vacuum extraction (Ventouse) and forceps should also be avoided.
As soon as the baby is delivered, a sample of blood from the umbilical cord should be collected to measure clotting factor levels. Injections into the baby’s muscle tissue and other surgical procedures, such as circumcision, should be avoided until the results of these blood tests are known.
After delivery, a carrier’s circulating clotting factor goes back down to her pre-pregnancy level and the chance of bleeding is at its highest.
Postpartum hemorrhage (PPH) is a major cause of maternal death and disability, especially in some parts of the world. Therefore carriers of hemophilia, particularly symptomatic carriers and women with hemophilia, should be cared for in an obstetric unit with close collaboration with the hemophilia team.
Certain precautions can be taken to reduce the risk of PPH: medications that keep the womb contracted can be given, and the placenta should be delivered by controlled traction of the umbilical cord. This is called “active management” for placenta delivery and has been shown to significantly reduce the risk of PPH.
Carriers are at risk of PPH for up to six weeks after childbirth and should be advised to see their doctor immediately if bleeding is excessive during this period. Treatment may be recommended as a preventative measure, especially in carriers with low clotting factor levels.
Symptomatic carriers and women with hemophilia usually don’t experience symptoms on a daily basis. They may, however, experience prolonged bleeding after an accident or medical intervention. When this happens, they must be treated in the same way as men with hemophilia. Several treatment options exist:
Desmopressin is a synthetic hormone that may help control bleeding in an emergency or during surgery. It can be injected intravenously, administered under the skin (subcutaneously), or given as a nasal spray.
Desmopressin does not work for everyone. All carriers of hemophilia A with clotting factor levels of less than 50% should have their response to the medication tested before they need to use it. Desmopressin is not effective in carriers of hemophilia B, as it does not raise levels of factor IX.
Desmopressin should not be used in some instances, for example in cases of head trauma and in women who are at risk of heart problems. Doctors should be familiar with the medication and how it should be used before prescribing it.
In carriers for whom desmopressin doesn’t work or is not recommended, infusions of clotting factor concentrates may be necessary when the risk of severe bleeding is high, such as before or during surgery.
The antifibrinolytic drugs tranexamic acid and aminocaproic acid are used to prevent the breakdown of blood clots in certain parts of the body such as the mouth and uterus. They can be used to control heavy menstrual bleeding and during minor surgeries and dental procedures.
Hormone therapy can be used to help control excessive menstrual bleeding. This includes combined hormonal contraceptives (which can be given orally, as skin patches, or vaginally) and the levonorgestrel-releasing intrauterine device/system (IUD or IUS).
Some women will continue to experience heavy menstrual bleeding, even with these medications. Though there are always some risks involved in surgery, procedures such as uterine (or endometrial) ablation and hysterectomy may be considered in rare circumstances.
Being a carrier of hemophilia can have a significant impact on a woman’s health and her academic, professional, and social life.
Excessive or prolonged menstrual bleeding can be especially difficult for young girls, who may isolate themselves from family and friends, miss days from school, or avoid social events due to pain, discomfort, or the fear of staining clothing. A girl’s self-image and confidence can be negatively affected if she experiences shame or embarrassment because of heavy menstrual bleeding.
Many carriers are not aware that their symptoms are abnormal and do not seek medical advice. Even when they do, caregivers are not always well informed about bleeding disorders and the right diagnosis may be overlooked. Furthermore, medical care for women is lacking in many countries around the world. There may be cultural taboos and obstacles preventing women from seeking help, particularly for menstrual problems.
Heavy and prolonged menstrual bleeding and pain can affect a woman’s sexuality and may cause problems in her marriage. Women may also need to take time off work each month because of heavy bleeding, which can impact their career choices or professional success.
Many carriers of hemophilia, like others at risk of passing on a genetic disease, also experience guilt. They may feel as though they should not have children because of the possibility of passing on a bleeding disorder, or having a daughter who must face this possibility in turn.
The prospect of marriage may be affected because men, or their families, may not accept the risk of having an affected child. If they do have children with hemophilia, the needs of that child can put pressure on all family members, including siblings.
Many hemophilia treatment centres can provide carriers with skilled and sensitive counselling. The professionals there can provide information and support to work through these complex feelings and to empower women to take charge of their condition and advocate for proper treatment. Building a support network of other women who are facing the same issues, through the hemophilia treatment centre or local patient organization, can be a great source of comfort.
© 2026 World Federation of Hemophilia